 The fact that the absolute value of specific entropy is unknown is not a problem because it is the change in specific entropy (Δs) and not the absolute value that is important in practical problems. The absolute entropy of a system is denoted by S and is defined based on the concepts of the third law of thermodynamics. Absolute values of Sm S m are what are usually tabulated for calculational use. However, the absolute value of the entropy of a physical system and the more general creative meaning and value of entropy were not clearly defined and explained by Clausius, which made the Clausius entropy a little mysterious and speculative. Entropy also describes how much energy is not available to do work. Entropy, denoted by the symbol S, is the thermodynamic property describing the amount of molecular randomness or disorder in a system. For example, the specific entropy of water or steam is given using the reference that the specific entropy of water is zero at 32☏. With the convention that the entropy of a pure, perfectly-ordered crystalline solid at zero kelvins is zero, we can establish the third-law value of the molar entropy of a pure substance at any temperature and pressure. Entropy is a measure of the disorder of a system. Entropy, the measure of a system’s thermal energy per unit temperature that is unavailable for doing useful work. Also, like enthalpy, the entropy of a substance is given with respect to some reference value. Write the mathematical equation that relates the standard entropy change in a chemical reaction to the absolute entropy of the. The third law of thermodynamics is based on the entropy. Like enthalpy, entropy cannot be measured directly. The determination of absolute entropy of solids, liquids and gases is possible. Entropy is represented by the letter S and can be defined as ΔS in the following relationships. This means that standard entropy is really just the absolute entropy for a substance at 298 K. Recall that standard entropies are standardized at 298 K. 
Entropy contained in a system, say in a mole of a pure substance, is a theoretical quantity that takes account of all heat transferred to it since the lowest atainable temperature, 0 K. looking at the behavior of heat capacities and entropy at lower and lower temperatures. Absolute entropy is the total amount of entropy acquired when a pure substance is warmed from absolute zero to a specific temperature. Entropy is sometimes referred to as a measure of the inability to do work for a given heat transferred. By definition, the change in entropy can be evaluated by measuring the amount of energy transferred. Because entropy tells so much about the usefulness of an amount of heat transferred in performing work, the steam tables include values of specific entropy (s = S/m) as part of the information tabulated. Entropy quantifies the energy of a substance that is no longer available to perform useful work.  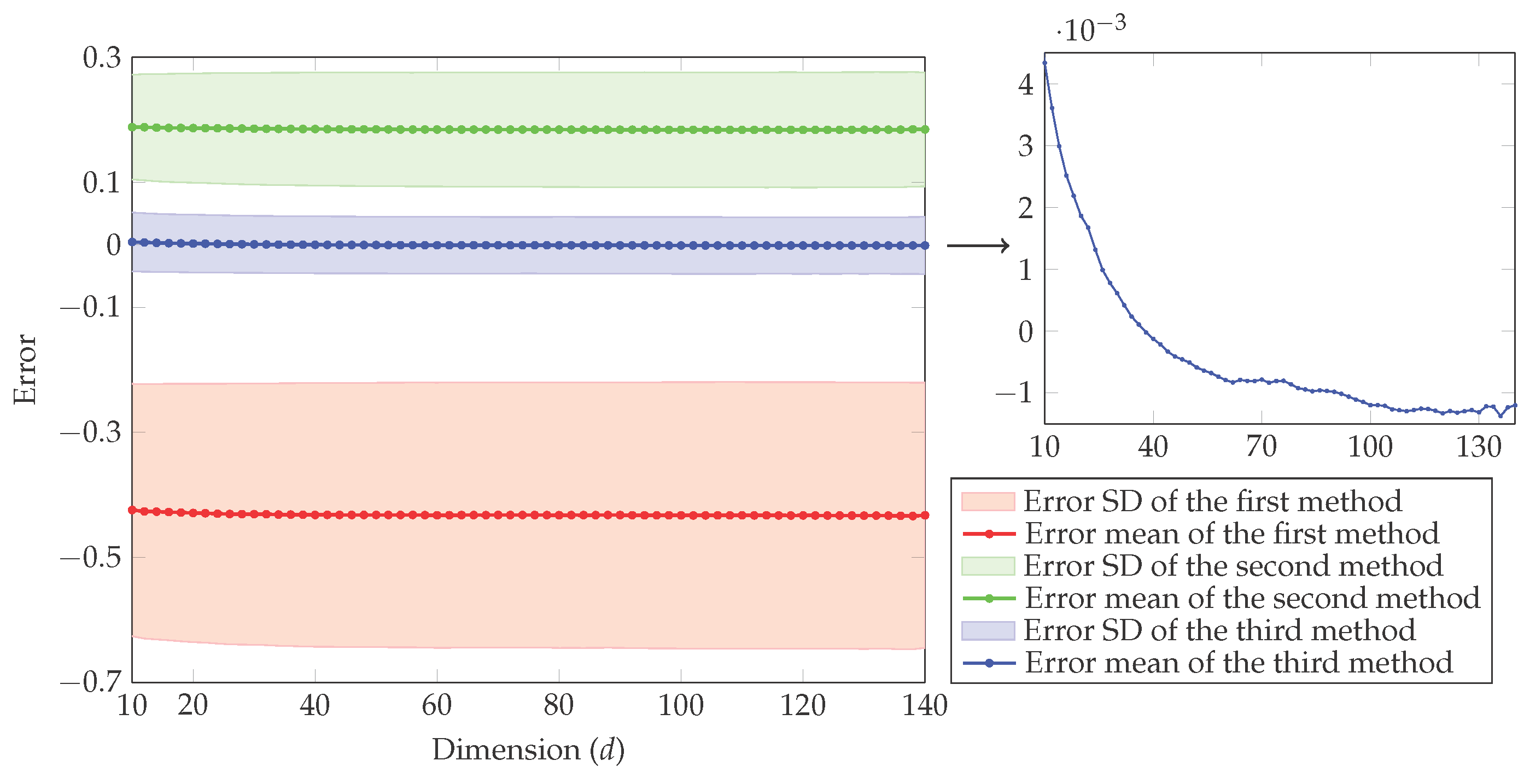
Because entropy is a property, changes in it can be determined by knowing the initial and final conditions of a substance. 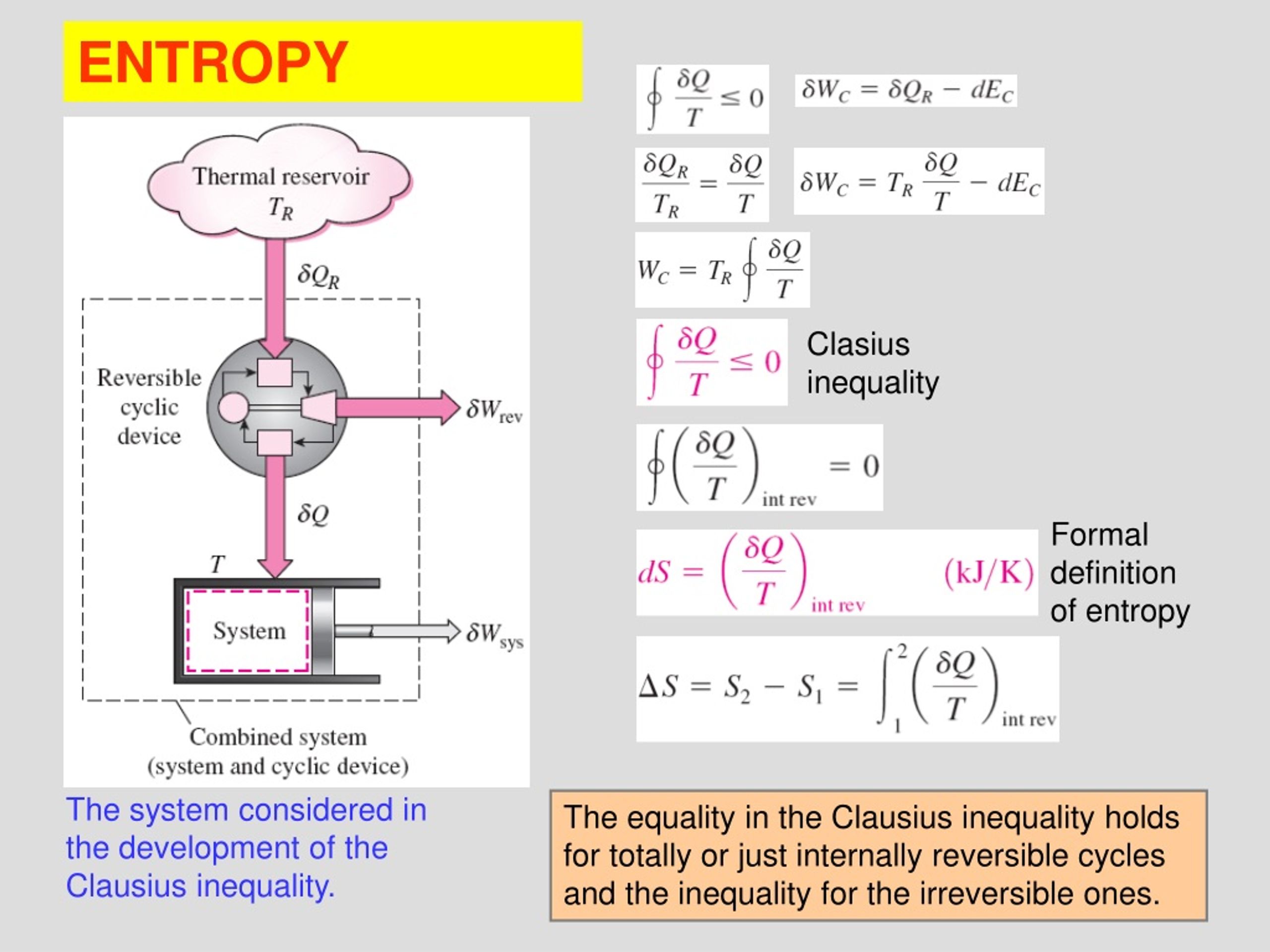
> Thermodynamics Directory | Heat Transfer DirectoryĮntropy Definition - Thermodynamic PropertiesĮntropy (S) is a property of a substance, as are pressure, temperature, volume, and enthalpy. The third law of thermodynamics has two important consequences: it defines the sign of the entropy of any substance at temperatures above absolute zero as positive, and it provides a fixed reference point that allows us to measure the absolute entropy of any substance at any temperature. Entropy Definition and Equation Thermodynamics 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
